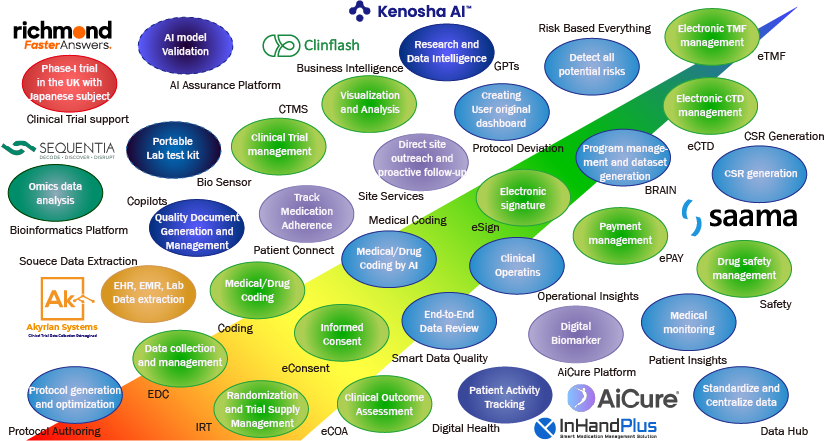
As illustrated below, our company offers a wide range of solutions that support the entire process of pharmaceutical and medical device development - from the early stages through regulatory submission. Our product lineup combines widely adopted core clinical development systems such as EDC and CTMS with cutting‑edge technologies designed to address longstanding challenges and shape the future of clinical operations.
The core solutions is the Clinflash® Cloud product suite from Clinflash, which covers nearly all operational needs in clinical development. In addition, through our partnership with Inventis, we provide globally standardized and properly managed devices for both patients and clinical sites, supporting services such as ePRO and eConsent device provisioning.
We also offer a range of advanced, AI‑driven solutions from companies including AiCure, Akyrian Systems, IhHandPlus, Kenosha AI, Saama, and Sequentia Biotech, helping our clients accelerate digital transformation and operational efficiency. Furthermore, to meet the growing demand for SaMD application development and distribution, we provide the Extra Horizon platform.
Beyond these IT solutions, our collaboration with Richmond Pharmacology in London enables us to support Phase I studies in the UK - including those involving Japanese participants - as well as provide access to facilities capable of conducting gene‑editing therapy trials. Through this expanded capability, we aim to further accelerate and streamline clinical development to address challenges such as drug lag and drug loss in Japan.
During COVID-19, with a growing demand for remote and decentralized solutions, AiCure was seen as a unique value because of its years of experience deploying and supporting patient-facing software. AiCure responded by developing compliant telemedicine solutions for its sponsors. Also, AiCure launched Clinical Site Services (CSS) to relieve technology burden at sites.
Today, AiCure continues to innovate and broaden its market presence as a platform for all things patient-facing. This includes eConsent, ePRO/eCOA, telemedicine, AI-driven patient engagement via two-way text, and of course, medication adherence and digital biomarkers.
AiCure has a proven record with dozens of clinical trials sponsored by the most prominent Big Pharma companies in the world. Its AI-platform has been deployed in over 40 languages. Importantly, AiCure has supported studies in all phases of drug development leading to FDA submissions, projects in post-marketing research, city- and state-wide deployments in public health, and large US Government and military contracts.
They created Akyrian Systems, and the result of years in R&D led to Akyrian’s flagship product, Source Data Extraction or SDE, a patented clinical data platform that is now poised to significantly reduce clinical trial duration and overall cost.
Services for Japanese customers are provided by the system within a cloud data center in Japan.
| Category/Product Name | Function |
|---|---|
| Cloud Data Solutions | |
| Clinflash® EDC | Data collection and management of Clinical trial data |
| Clinflash® IRT | Randomization and Trial Supply Management |
| Clinflash® Coder | Medical and Drug Coding |
| Clinflash® BI | Data visualization, analysis and central monitoring |
| Digital Cloud Solutions | |
| Clinflash® eCOA | Electronic Clinical Outcome Assessment |
| Clinflash® eCTD | Electronic Common Technical Document |
| Clinflash® ePAY | Clinical Trial Electronic Payment |
| Clinflash® eSign | Clinical Trial Electronic Signature |
| Clinflash® eConsent | Electronic Informed Consent |
| Project Management Cloud Solution | |
| Clinflash® CTMS | Clinical Trial Management System |
| Clinflash® eTMF | Electronic Trial Master File |
| Pharmacovigilance Cloud Solution | |
| Clinflash® Safety | Pharmacovigilance Safety Database |
By breaking the recurring barriers involved in building a MedTech solution, we help get digital health products to the people who need them as quickly as possible, improving the quality of healthcare and making a lasting impact.
Specific examples include tracking medication, detecting scratching and tremors, recording each meal and calculating calories, and analyzing the effects of food. It is expected to be applied to clinical trials and research, as well as dietary advice and DCT.
Furthermore, Inventus offers “Inventus Connect,” a global eSIM platform available in more than 150 countries that greatly simplifies device connectivity management in clinical trials. Its carrier agnostic network architecture enables stable cross border data communication, supporting operational efficiency and cost optimization. In addition, the Inventus Device Management (IDM) software, purpose built for clinical trials, provides centralized control of device setup, OS updates, and security management, delivering a level of compliance and operational efficiency that general commercial devices cannot match.
This product has several features, including its low price (please inquire about the price), fast data updates (you can search for notifications from the previous day), and the display of the type of search being performed based on the user's prompt. In addition, since the LLM is tuned specifically for regulatory information, the hallucination is minimized and you can quickly access the desired information.
Richmond Pharmacology has over 20 years of experience conducting over 100 Phase-I trials in London targeting Japanese volunteers, and is a very unique CRO with the largest Japanese database (24,000 people) of any facility based in Europe, and has a track record of being used by many clients, particularly those planning international joint clinical trials. In addition, recently, trials targeting specific patient cohorts have accounted for approximately 60% of the total number of trials, and the company is also focusing on clinical trials in the fields of gene silencing and gene editing. While accurately grasping the needs of the times, the company is working hard to accelerate research using adaptive designs and provide "Faster Answers."
Saama uses artificial intelligence (AI), along with advanced analytics, to automate key clinical development processes and surface actionable insights. This allows customers to accelerate their time to market and eliminate manual, resource-intensive processes.
Saama accomplishes this mission through the combination of our strong portfolio of SaaS-based solutions, ready for deployment out-of-the box, as well as our ability to work with your organization to build and deploy custom AI and advanced analytics-based solutions. This combination of capabilities means we can cover the entire spectrum of clinical development and commercialization.
| Category/Product Name | Function |
|---|---|
| Data centralization and standardization platform | |
| Data Hub | Data integration platform for all applications below |
| AI assisted applications for Clinical development processes | |
| Smart Data Quality | AI assisted End-to-End data review |
| Operational Insights | AI assisted Operational review |
| Patient Insights | AI assisted patient journey review |
| Source to Submission | AI assisted SDTM transformation |
| BRAIN | Integrated program and dataset management environment |
| Document Generation platform | |
| Protocol Authoring | Protocol generation and management |
| CSR Authoring | CSR generation and management |
For the pharmaceutical industry, applications include designing personalized medicine and discovering new biomarkers using Omics data and clinical data (RWD and clinical trial data), but we believe that it can also be applied to agriculture, livestock farming, environmental science, and more.